Deuterium Atom Paradox:
More Gravitational, Less Energetic Only wave theory's two-swirl formation
provides a suitable explanation for the structure of atoms
and all their isotopes
Only wave theory's two-swirl formation
provides a suitable explanation for the structure of atoms
and all their isotopes
[The
Hydrogen Atom]
Until now, deuterium atoms were thought
to be more energetic than hydrogen atoms. However, this accepted
fact has been debunked: off the frozen coast of Norway, hydrogen
has been found to lose energy and turn into deuterium.
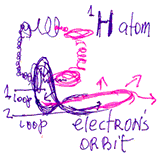 
Despite the fact that the deuterium atom
is less energetic than the hydrogen atom, it nonetheless remains
the preferred and most effective material with which to build
the neutron bomb. I contend that the reason for this seeming
paradox is that the hydrogen atom is more dispersed. In other
words, its energy is organized in a loose, cloudy neutron
formation, and its pushing properties are greater than its
gravitational forces. On the other hand, the gravitational
forces in the deuterium atom are superior. Consequently, its
energy is stored in a compact manner, which makes it a more
exploitable source of energy than the mercurial hydrogen atom,
even though it is less energetic.
* * * * *
As an aside, the deuterium atom has a
poisoning effect on organic formations, as it obstructs their
access to highly-energetic hydrogen atoms.
Back to Top
Dr. Chaim Tejman, Copyright©
2003. All rights reserved.
[Index]
[Introduction]
[Summary] [Book]
[Wave Formation] [Photons]
[Gravitation] [Time]
[Life] [Cancer]
[Fundamental Force] [Gender/Why
Sex?] [Sexual Reproduction]
[Schrodinger & Heisenberg]
[Creation] [Supernova]
[Dark Matter & Astronomy]
[Speed of Light] [Cloud
Formations] [Natural Disasters]
[Global Warming] [Thermodynamics]
[Backward Time] [Quantum
Mechanics] [Compton Effect]
[Equations] [Predictions]
[Academic Correspondences] [Contact]
[Links] [Mysteries] |